Silicon has been a substance of great importance for humanity since the days our ancient ancestors first discovered that this can be fashioned into cutting tools due to its properties that make it strong yet able to be fractured fairly easily. Silicon is a metalloid chemical element (considered a crystalline) which has conductive properties making it valuable for technological research. Recently scientists out of Stanford University have discovered that its conductive properties can improve the efficiency of lithium-ion batteries which are currently used for a variety of functions including those used in hybrid cars and some models of airplanes.
What are the benefits of Lithium-Ion Batteries?
Lithium-Ion batteries are commonly known to most people as “rechargeable batteries” which is one of the main reasons these are increasing in popularity to save the cost of having to recycle the components of a batteries shell. Previously nickel-cadmium was used to create batteries however chemists discovered in the 1990’s that lithium ion is a superior substance to use for this function because it can store almost twice the charge of its predecessor. With our ever increasing concentration on improving energy supplies it appears as if silicon technology could be implemented in the next generation of energy storage systems such as batteries.
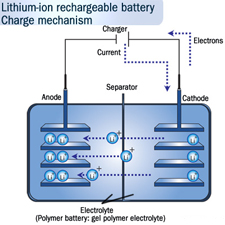
Silicon Technology:
Chemical engineer Zhenan Bao of Stanford University understands the growing need of energy resources and how more efficient storage devices are required to enhance our ever increasing use of portable technology. Bao and his colleagues came to the realization that silicon is the best natural resource to help improve both the cost and efficiency of battery life while also being environmentally friendly. Being a naturally occurring element with almost no negative environmental side effects, silicon is perfect for implementation in batteries which were once known for being potentially harmful in terms of environmental waste byproducts.
The latest studies into the combination of silicon and lithium have shown that the two elements together can provide approximately 400% expansion when used towards energy storage for a battery. Unfortunately there is a side effect to this expansion in that fractures can occur in the electrical contact when batteries are recharged or discharged. Currently this is being resolved by Stanford researches by applying a hydrogel on silicon nanoparticles in a technique referred to as in situ synthesis polymerization.

Nanoscale Architecture:
Bao attributes the new generation of batteries his colleagues are researching not only to the materials used in them, but also to the design they have created for the placement of materials and nanoparticles creating an electrochemical improvement over older versions. This has become possible using electron microscopes to analyze how the spaces between silicon nanoparticles can be properly optimized to fit into the spaces between the hydrogel used thus allowing a more efficient expansion capability when lithium is used.
One of the issues encountered in this study however was the fact that hydrogel and lithium have a potential of sparking fires due to the main component in hydrogel being water. The only way to counter this currently is by removing the water particles after the hydrogel has been set in place to minimize the chances of having lithium ignite when it comes in contact with water. Along with this there are a few other technical issues which have been discovered in the creation of this new battery, however Yi Cui an associate professor at Stanford holds onto his optimism that this new battery can be manufactured for commercial use soon once all the kinks are worked out of the technical design of it.
![]()
